For Sponsors
A Site Partner Built for Quality, Speed, and Diversity
GCSA-certified, ICH-GCP E6 (R3) compliant. Miami HQ with Kendall trial-ops office. Top-5 cardiovascular trial site in the Southeast US.
Why Amavita
Six reasons sponsors choose Amavita Research as their Miami site partner.
IAOCR GCSA Certified
Independently accredited site quality and competency standards.
ICH-GCP E6 (R3) Compliant
Operating to the most current Good Clinical Practice guidelines.
Blue-Chip Track Record
Active partnerships with Alnylam, Pfizer, JK07 Bio, VASA Therapeutics and others.
Top-5 CV Site, Southeast US
Recognized cardiovascular trial performance across the region.
Diverse Miami Population
Hispanic, Caribbean, and multi-ethnic enrollment depth.
Dedicated BD Contact
Responsive, single point of contact for feasibility and contracts.
A Senior PI with a track record of FDA-pivotal device trial leadership
Our Senior Principal Investigator Dr. William W. O'Neill, MD, FACC has served as National Principal Investigator on the Edwards PARTNER, Protect II, and Protect IV trials — a track record of FDA-pivotal device trial leadership matched by few investigators in the United States.
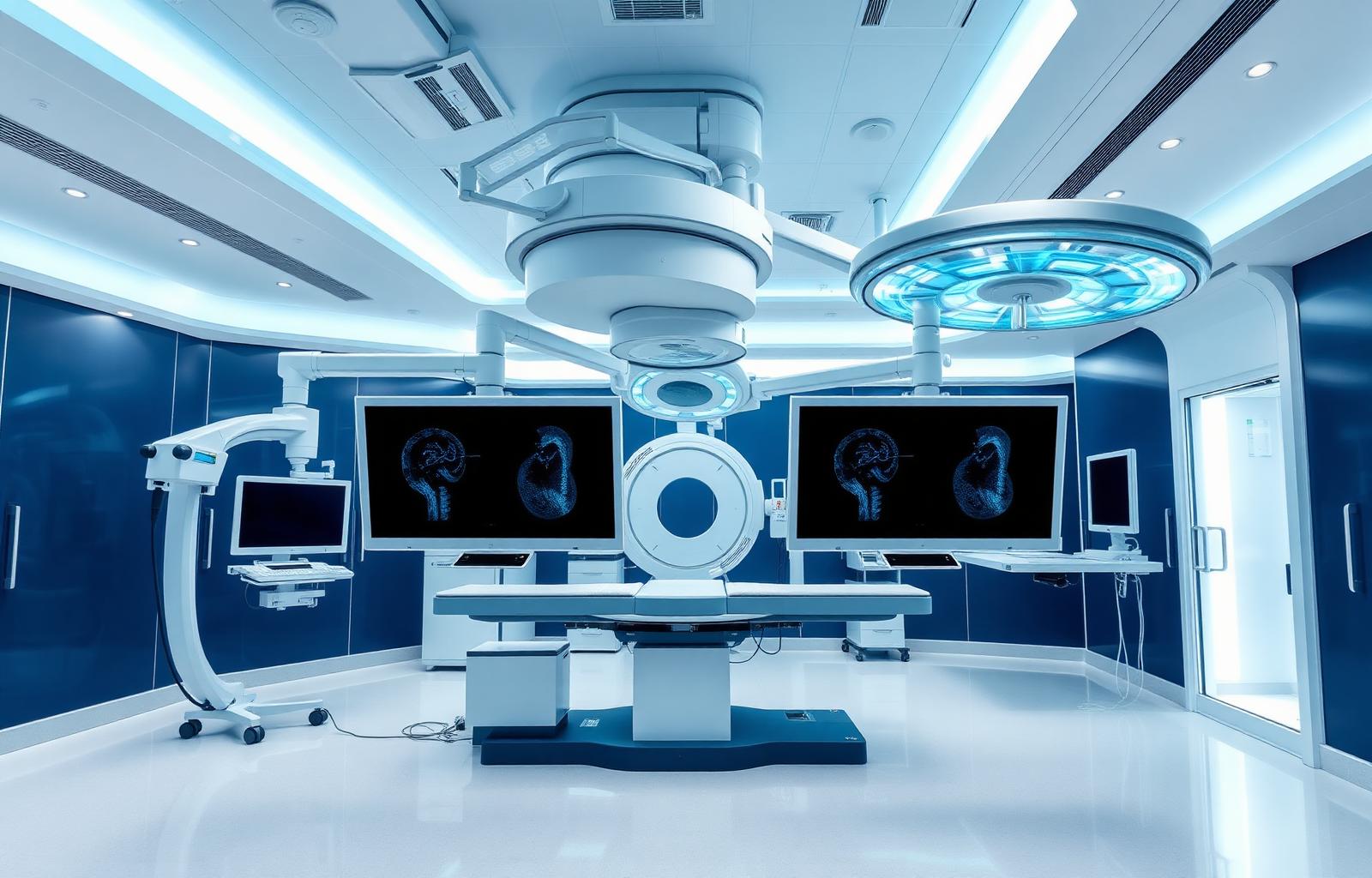
South Florida's only dedicated cardiovascular ambulatory surgery center
Sponsors partnering with Amavita Research gain access to South Florida's only dedicated cardiovascular ASC — enabling complex device and structural-heart studies, same-day procedures, and a controlled outpatient setting that streamlines protocol execution and reduces hospital-based variability.
ASC Capabilities
Procedures supported on-site
Complex device, structural-heart, and interventional studies executed in a controlled outpatient setting — reducing hospital-based variability.
Cardiology
- Cardiac Catheterization
- Cardiac Mapping & Ablation
- Coronary Intervention
- Implantable Cardiac Monitors
- Pacemakers / ICDs / Bi-Ventricular Devices
- Peripheral Artery & Vein Angiography & Intervention
Interventional Radiology
- Dialysis Access Management
- Genicular / Hemorrhoidal / Prostate Artery Embolization
- Kyphoplasty
- Spinal Cord Stimulator Implantation
- Uterine Fibroid Embolization
Technology

Capabilities that move your study forward
From feasibility through close-out, our team operates as a true extension of yours — bringing deep cardiometabolic experience and the infrastructure to execute complex protocols.
Therapeutic Expertise
Deep experience across cardiology, heart failure (HFrEF/HFpEF), hypertension, ACS, vascular and metabolic studies — Phase 1 through Phase 3.
Modern Recruitment Technology
EHR-integrated patient identification, predictive screening, and a digital pre-screening funnel that accelerates enrollment.
Trained, Bilingual Staff
Board-certified PI, study coordinators, and regulatory specialists fluent in English and Spanish.
Miami-Area Patient Diversity
Access to one of the most ethnically and clinically diverse patient populations in the United States.
Trusted by leading sponsors
- Alnylam Pharmaceuticals
- Pfizer
- JK07 Bio
- VASA Therapeutics
- Pharmacosmos
- Cardiovascular Systems
- Boehringer Ingelheim
- Novartis
- AstraZeneca
- Bristol Myers Squibb
- Merck
- Bayer
- Eli Lilly
- Amgen
- Sanofi
- Gilead
16+ pharmaceutical and biotech partners — selection shown
Track Record
Performance you can rely on
Sponsors return to Amavita Research because we deliver clean, on-time data — and a patient experience that drives retention.
- 10 active trials, growing to 18–25
- 16+ pharmaceutical and biotech sponsors
- Consistently meets or exceeds enrollment targets
- Audit-ready quality systems and SOPs
- Rapid IRB submission and contract turnaround
- SCRS Global Summit participant
Business Development
Nereisy Alonso, Sr CRC
Business Development Director
Schedule a Sponsor ConsultationFAQ
Frequently Asked Questions
Quick answers about our certifications, leadership, and sponsor partnerships.