News
Coronary Stent Procedure Performed at Miami-Dade County's First Cardiovascular Ambulatory Surgery Center
A coronary stent procedure performed at Amavita's Kendall ASC — the first cardiovascular ambulatory surgery center in Miami-Dade County, with same-day discharge.
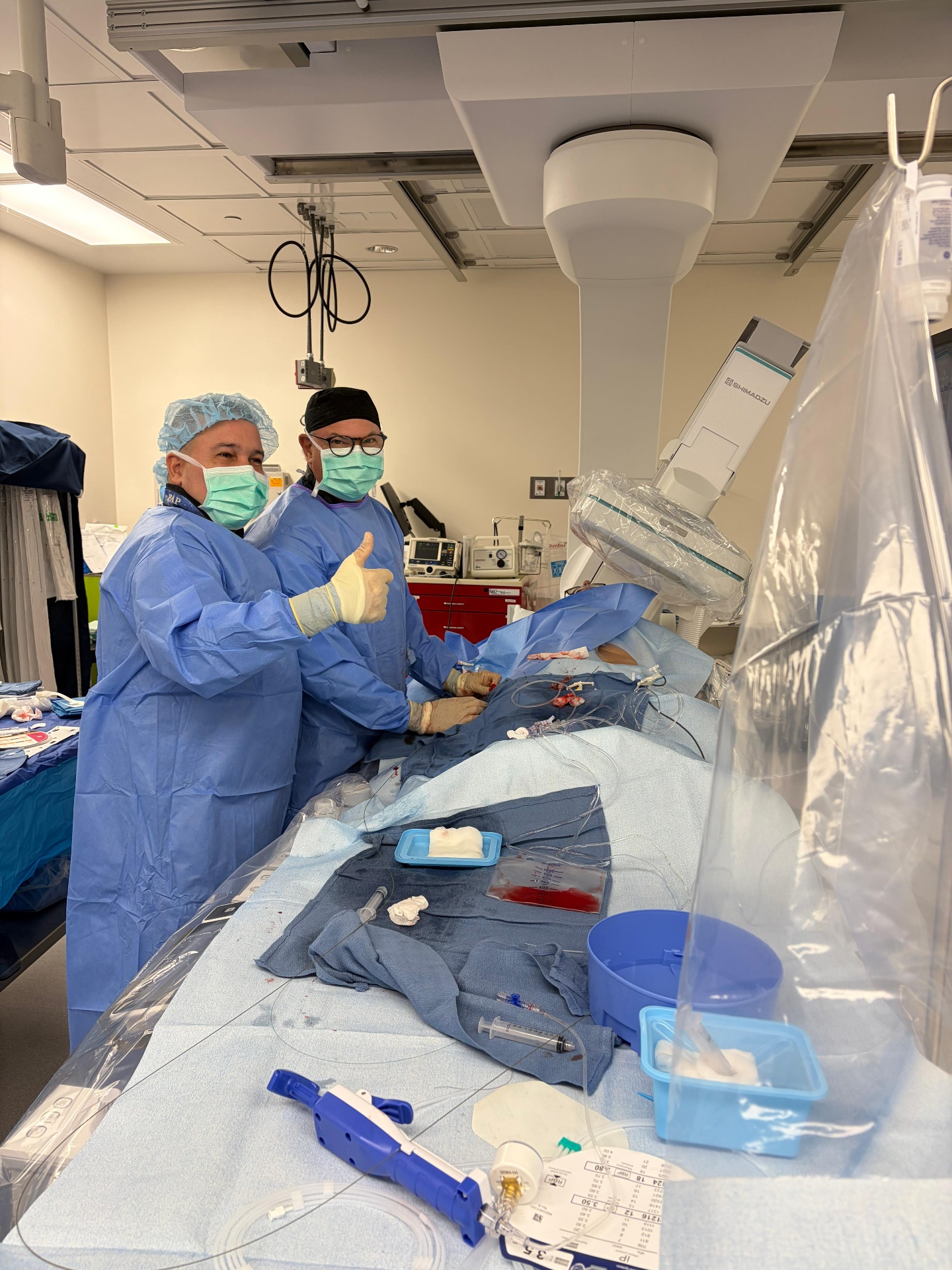
Miami, FL — May 13, 2026. Earlier today, Amavita Heart and Vascular Health® performed a coronary stent procedure at Amavita's Kendall ASC — the first cardiovascular ambulatory surgery center (ASC) in Miami-Dade County. The percutaneous coronary intervention (PCI) was led by Dr. Pedro Martinez-Clark, Lead Investigator at Amavita Research Services LLC. The same facility houses our cardiovascular Phase 1 clinical-trial unit.
The patient was discharged the same day, fully recovered, and walked out of the facility without an overnight hospital stay — the central clinical promise of outpatient PCI.
Why this matters
For decades, coronary stenting in the United States was performed exclusively in hospital cath labs, with most patients admitted overnight for observation. In 2020, the Centers for Medicare & Medicaid Services (CMS) added PCI procedures to the ASC-payable list as part of the 2020 OPPS/ASC final rule (CMS-1717-FC), recognizing that select, well-screened patients could safely undergo coronary intervention in an outpatient setting and go home the same day.
Since 2020, ASC-based PCI has expanded across the country — but Miami-Dade County did not have a dedicated cardiovascular ASC until Amavita's Kendall facility was licensed and equipped for cardiovascular procedures. Today's PCI was performed at that facility — the first cardiovascular ASC in the county.
It is the result of three things coming together at the same facility:
- A purpose-built cath-lab suite at Amavita's Kendall ASC, fluoroscopy-equipped and outfitted to current interventional cardiology standards.
- Same-day recovery infrastructure — an eight-bed recovery suite where post-procedure patients can be observed before discharge, the same suite used by our Phase 1 trial unit.
- A board-certified interventional cardiology team led by Dr. Pedro Martinez-Clark, with Dr. William O'Neill as Senior PI on cardiovascular research, and Dr. Hans Rutzen-Lopez on the electrophysiology side.
What this signals to clinical-trial sponsors
Beyond the clinical milestone, today's procedure is a proof point for sponsors evaluating Amavita as a cardiovascular trial site. The same combination — interventional capability, on-site recovery beds, an IRB-aware research operation — is exactly what device sponsors need for early-feasibility, first-in-human (FIH), and pivotal cardiovascular device trials.
If your team is running a cardiovascular device trial — coronary stent, drug-coated balloon, intravascular imaging, structural heart, or peripheral intervention — and you need a site that can execute the procedure under both commercial and research protocols in the same building, with same-day discharge as a routine workflow rather than an exception, we should talk.
Amavita Research holds IAOCR GCSA recertification through 2028, making it one of the few cardiovascular clinical-trial sites in the United States with formal good-clinical-site-accreditation. Our broader cardiovascular trial-site capabilities are documented at /capabilities and /certifications, and our sister organization bioaccess® brings 18+ years of medical-device FIH trial execution in Latin America.
About the procedure
Out of respect for patient privacy, we are not disclosing patient demographics or clinical history. The case was selected and consented under Amavita's standard interventional cardiology screening protocol. Same-day discharge was planned in advance and executed without complication.
Press contact: For sponsor inquiries and press follow-ups about cardiovascular Phase 1 unit capability or ASC-based PCI at Amavita, contact: researchadmin@amavita.health